Translate this page into:
Whole-body Positron Emission Tomography (WBPET) Scan in All Patients of Breast Cancer: Is This the Way Forward?

*Corresponding author: M. G. Vishnoi, Department of Nuclear Medicine, Command Hospital, Kolkata, West Bengal, India. mgv_2006@rediffmail.com
-
Received: ,
Accepted: ,
How to cite this article: Vishnoi MG, Bandyopadhyay A, Gupta A, Rathore A, Sharma A, Pandya T, et al. Whole-body positron emission tomography (WBPET) scan in all patients of breast cancer: Is this the way forward? Indian Cancer Awareness J 2022;1:15-20.
Abstract
Objectives:
Globally, breast cancer is the second most frequently diagnosed malignancy just behind lung cancer, accounting for over 2 million cases each year. It is the second most commonly diagnosed cancer worldwide and the most common malignancy among females in India. The role of whole-body positron emission tomography (WBPET) in all cases of breast cancer is still not clear. This pilot study was undertaken to study whether there is any change in the management of breast cancer using WBPET scan in all patients of breast cancer irrespective of the stage?
Materials and Methods:
We retrospectively reviewed the data of 43 patients with invasive breast cancer who were newly diagnosed at Malignant Disease Treatment Centre at a tertiary care centre in East India. The duration of the study was between June 2020 and March 2021. Patients underwent liver function test, mammography, breast ultrasound, bone scan, and tru-cut needle biopsy to confirm the presence of breast cancer and 18F-fluoro-Dglucose (FDG) positron emission tomography/computed tomography (PET/CT) was studied in all 43 patients before the initiation of any therapy. The primary objective was to find out whether doing a WBPET upstaged the disease and changed our management. The secondary objective was to determine whether hormonal status (oestrogen receptor [ER], progesterone receptor [PR] and human epidermal growth factor receptor 2 [Her2] Neu) and Breast Imaging-Reporting and Database System (BIRADS) score staging had any correlation with metastasis.
Results:
A total of 43 patients with newly diagnosed breast cancer were studied. The number of patients in Stage IIA (3/43), Stage IIB (4/43), Stage III A (17/43), Stage IIIB (13/43), and Stage IV was (6/33). Out of total 43 patients, in 10 (23%) patients, there was a change in treatment on doing an upfront WBPET scan. There was no correlation between ER, PR, or Her2 Neu positivity or negativity on the upstaging of the disease and change in treatment. Standardised uptake value (SUV) uptake of the primary lesion >7.2 was associated with upstaging and change of treatment.
Conclusion:
The use of 18F-FDG PET/CT has been shown to bring change in treatment in almost 20% of patients with breast cancer. Although the role of other variables in breast cancer such as hormone status, SUV of the primary lesion, and grade of lesion on BIRADS has also shown some association with change of treatment.
Keywords
WBPET
ER
PR
INTRODUCTION
Globally, breast cancer is the second most frequently diagnosed malignancy just behind lung cancer, accounting for over 2 million cases each year.[1] It is also the leading cause of cancer death in women worldwide. In the United States, breast cancer is the most common female cancer and the second most common cause of cancer death in women.[2] It is the second most commonly diagnosed cancer worldwide.[1]
Over the years, cervical cancer had been the most common malignancy in India but over the past decade, the number of breast cancers has been increasing rapidly and is now the most common malignancy among females in India. Hence, roughly, in India, for every two women newly detected with breast cancer, one woman is dying of it. In 2018, 162,468 new cases of breast cancer were diagnosed, representing 27.7% of all new cancers among Indian women and 11.1% of all cancer deaths.[3] Although the number of patients presenting with Stage IV is only about 10% in the United States, in India, approximately 29–52% of Indian women presenting at Stage III and Stage IV in 6–24% of women with breast cancer.[4]
Since the presentation of breast cancer in India is at a more advanced stage and at a relatively younger age, the question we need to answer is whether we need to bring change in our diagnosing, staging, and management of breast cancer in India?
Once a diagnosis of breast cancer is established, it is important to accurately define the initial extent of the disease. The stage of the disease has the most important bearing on the management of the disease. The various imaging techniques used in diagnosis and staging of the disease are classic mammography, breast ultrasound (USG), magnetic resonance imaging (MRI) breast, bone scan, computed tomography (CT) scan, and positron emission tomographyCT (PET-CT) scan depending on the clinical profile of disease and availability.
In cases of patients with Stage IIIA or higher disease, patients with inflammatory breast cancer regardless of whether symptoms are present or not, whole-body PET-CT is indicated. In places where whole-body PET (WBPET) CT is not available, contrast-enhanced CT (CECT) of chest, abdomen, and pelvis along with bone scan is a reasonable alternative.
Most of the guidelines including NCCN do not indicate PET scan with apparently early Stage I or II breast cancer and those with operable Stage III breast cancer. However, there has been a growing trend among oncosurgeons so as to get a WBPET scan in all cases of breast cancer. The reasons may be many, but the most relevant reason could be that of upstaging of the disease may change the treatment. According to many, it is a one-stop shop and after a WBPET scan, the requirement of CECT/bone scan/MRI breast/USG axilla even the metabolic work up all are taken care of. Moreover, it appears reasonable test to perform in cases of suspicious or equivocal findings on CT or MRI and for women with locally advanced disease. This helps the patients both economically as well as saves the patients from multiple hospitals visit.
Hence, the question which needs to be addressed is has the time come when WBPET be done in each and every case of breast cancer irrespective of its stage? There have been studies that have shown the advantage of using WBPET in cancer patients, but to date, none of the guidelines have suggested these changes. Hence, to answer this question, we have tried to have a retrospective analysis of data of 43 consecutive patients who underwent WBPET scan at the time of diagnosis in our centre.
What is a PET scan?
PET has been an important diagnostic tool in staging many malignancies. The most widely used US Food and Drug Administration (FDA)-approved PET radiotracer, 2-deoxy-2-(18F) fluoro-D-glucose (FDG), is based on glucose metabolism. FDG is transported into the cell through glucose transporters, but once inside the cell unlike glucose, FDG is not metabolised. It is irreversibly phosphorylated by hexokinase and trapped within the cell. Glucose is taken up more by cancer cells compared to normal cells and helps in visualisation of tumours.
FDG uptake in breast cancers depends on the aggressiveness of the tumour in the form of mitotic activity, grade, and tumour cell density include mitotic activity index.[5] Hence, FDG uptake is greater with triple-negative breast cancer and human epidermal growth factor receptor 2 (HER2) positivity, and lower with luminal A subtypes. It is also used as a prognostic marker for response to therapy when neoadjuvant chemotherapy is used in breast cancer.[6] It is a problem-solving method when the results of conventional imaging are equivocal.[7] However, one also needs to understand there are wide spectrum hypermetabolic lesions including benign and other malignant lesions in the breast apart from breast cancer.
MATERIALS AND METHODS
Study patients
We retrospectively reviewed the data of 43 patients with invasive breast cancer who were newly diagnosed at Malignant Disease Treatment Centre at a tertiary care centre in East India. None of these patients had received any prior chemotherapy or had any feature suggestive of bilateral breast disease, N3c disease, or metastatic disease at the time of diagnosis. The duration of the study was between June 2020 and March 2021.
Patients with invasive breast cancer who were diagnosed with excisional biopsy were excluded from this study, because of the non-visualisation of the primary tumour. Patients having any contraindication for WBPET scan (like an anaphylactic reaction to any contrast or claustrophobia) were excluded from the study.
Most patients underwent liver function test, mammography, breast USG, and tru-cut needle biopsy to confirm the presence of breast cancer. A bone scan was done in patients who had a symptomatic bone pain or raised alkaline phosphatase levels. The 18F-FDG PET/CT was studied in all 43 patients before the initiation of any therapy.
We reviewed the medical record of each patient for clinical information including age at diagnosis and other pathological parameters were including the tumour size, lymph node (LN) metastasis, and hormone receptor status. Staging of the disease was done using the AJCC cancer staging manual 8th edition.
The primary objective was to find out whether doing a WBPET upstaged the disease and changed our management. The secondary objective was to determine whether hormonal status (oestrogen receptor [ER], progesterone receptor [PR] and Her2 Neu), and Breast Imaging-Reporting and Database System (BIRADS) score staging had any correlation with metastasis.
Statistical analysis was done using Chi-square test. This test was used for the testing relationship between categorical variables. All statistical tests, multivariate analysis, and data visualisation were done in R version 3.6.3. The Institutional Ethical Committee approved the study protocol and the study was conducted according to the Helsinki Declaration. Written consent was obtained from all study participants.
RESULTS
A total of 43 patients with newly diagnosed breast cancer were studied. The number of patients in Stage IIA (3/43), Stage IIB (4/43), Stage III A (17/43), Stage IIIB (13/43), and Stage IV was 6/33. In Stage IIA, out of three patients, one got upstaged but there was no change in the treatment modality. In Stage IIB, out of the four patients, two got upstaged and in fact one had a liver metastasis diagnosed leading to the change of treatment from curative intent to palliative intent. Rebiopsy from the liver lesion was suggestive of metastasis from the breast lesion. In Stage IIIA, out of 17 patients, four were upstaged and three had their treatment changed. The treatment change was in the form of the addition of NACT or palliative chemotherapy depending on the presence of metastasis. The maximum number of patients who had their disease upstaged was in Stage IIIB, where out of 13 patients, six were upstaged to metastatic disease and were further treated with palliative intent. In the six patients who were already Stage IV due to liver/skeletal/lung metastasis had more metastatic lesions discovered on WBPET, they continued with their palliative chemotherapy/hormonal therapy [Table 1].
| Stage of disease | Number of patients | Upstaging | Change in treatment |
|---|---|---|---|
| IIA | 3 | 1 | 0 |
| IIB | 4 | 2 | 1 |
| III A | 17 | 4 | 3 |
| III B | 13 | 6 | 6 |
| IV | 6 | 0 | 0 |
The various variables which have been studied whether they have a bearing on change of treatment are depicted with or without change in treatment are listed in [Table 2]. The average age of patients studied was 49 years, age-wise distribution of patients is depicted in the chart [Graph 1].
| S. No. | Characteristics | Change in t/t | No change in t/t |
|---|---|---|---|
| 1 | IDC (NOS) | 9 (10) | 32 (33) |
| 2 | Invasive lobular carcinoma | 1 (10) | 0 (33) |
| 3 | DCIS | 0 (10) | 1 (33) |
| 4 | ER+ | 3 (10) | 19 (33) |
| 5 | ER- | 7 (10) | 14 (33) |
| 6 | PR+ | 4 (10) | 17 (33) |
| 7 | PR- | 6 (10) | 16 (33) |
| 8 | Her2 Neu+ | 1 (10) | 1 (33) |
| 9 | Her2 Neu- | 9 (10) | 32 (33) |
| 10 | Grade III | 3 (10) | 14 (33) |
| 11 | Grade II | 1 (10) | 11 (33) |
| 12 | Grade I | 6 (10) | 8 (33) |
| 13 | BIRADS V | 7 (10) | 8 (33) |
ER: Oestrogen receptor, PR: Progesterone receptor, HER2: Human epidermal growth factor receptor 2, BIRADS: Breast imaging-reporting and database system
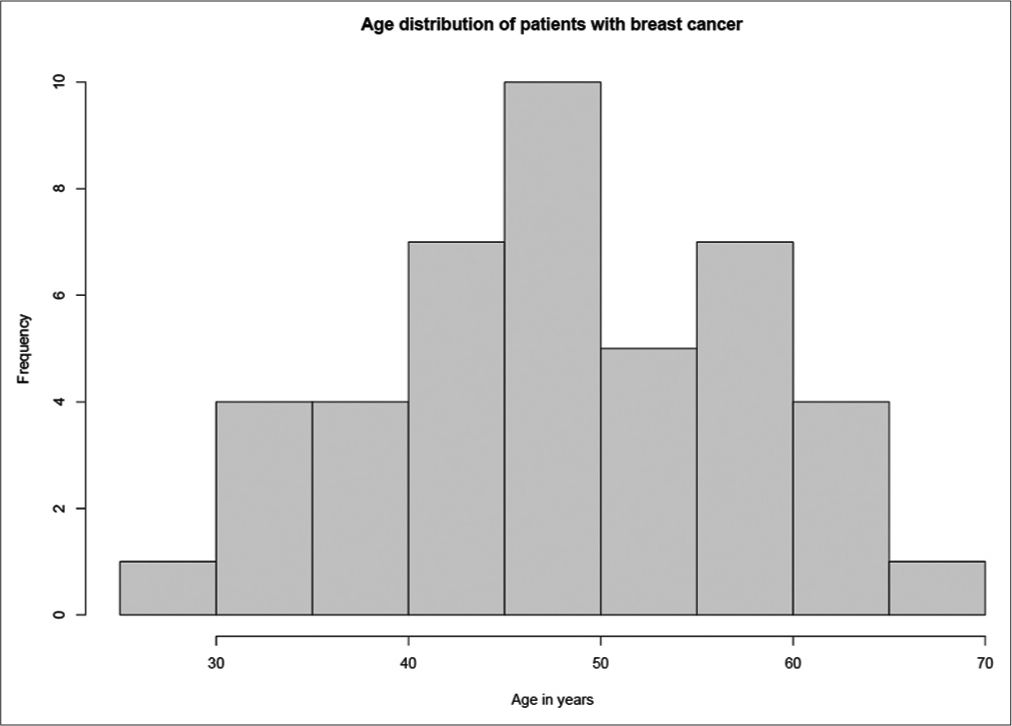
- Age-wise distribution of patients is depicted in the chart.
Out of these entire 43 patients, there was one male patient with carcinoma breast and the rest were all females. Out of total 43 patients, in 10 (23%) patients, there was a change in treatment on doing an upfront WBPET scan. The reason for the change in treatment was mainly due to the disease being detected to be metastatic on evaluation by WBPET. There was no correlation between ER PR or Her2 Neu positivity or negativity on the upstaging of the disease and change in treatment. This is depicted in the Forest plot graph [Graph 2].
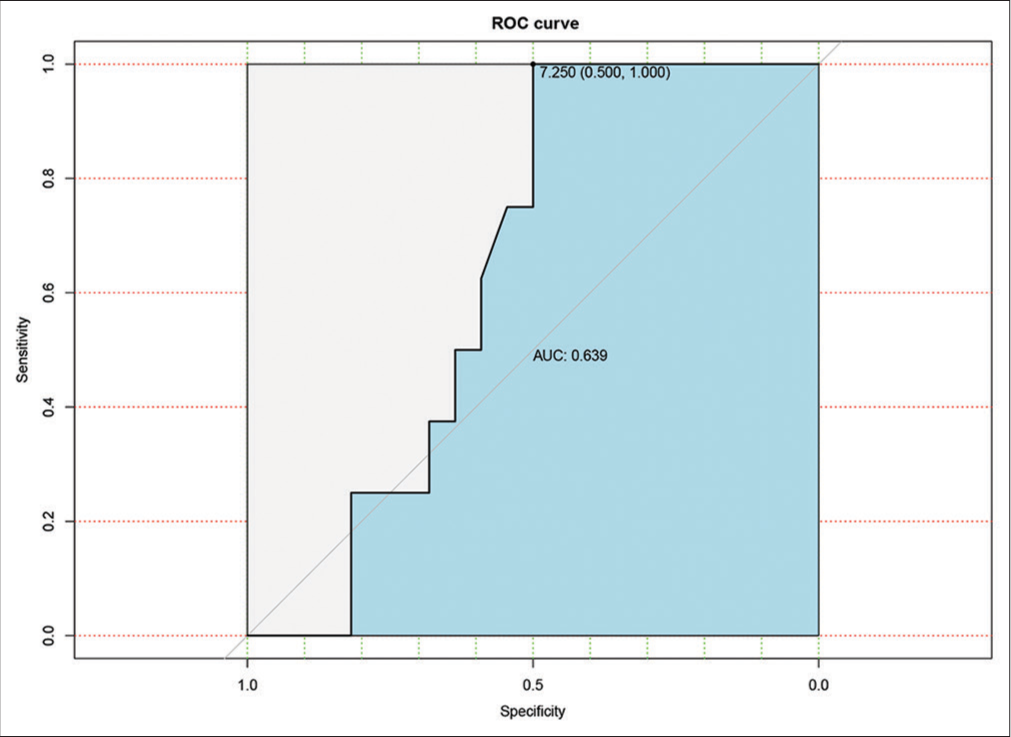
- Receiver operating characteristics curve for the ability of the standardised uptake value (SUV) of the primary lesion to identify patients requiring a change of treatment after positron emission tomography/computed tomography. The area under curve = 0.639. At SUV of 7.25, sensitivity and specificity are 100% and 50%, respectively.
Although the number of patients studied was limited, there has been a trend of change of treatment with rising BIRADS score compared to ER, PR, and Her2 Neu status [Table 3]. The standardised uptake value (SUV) of the primary lesion is always suggestive of the aggressiveness of the disease. However, just on the basis of SUV, it is very difficult to suggest whether the disease is metastatic or not. To address this question of whether a particular value of SUV is more suggestive of metastatic disease, we analysed our data. The outcome of receiver operating characteristic (ROC) curve for the ability of the SUV of the primary lesion to identify patients requiring a change of treatment after PET-CT. The area under curve (AUC) = 0.639. At SUV of 7.25, sensitivity and specificity are 100% and 50%, respectively [Graph 2].
| Parameter | Status | Change in treatment | P-value | |
|---|---|---|---|---|
| No | Yes | |||
| ER | Negative | 8 | 5 | 0.35 |
| Positive | 18 | 4 | ||
| PR | Negative | 9 | 5 | 0.43 |
| Positive | 18 | 4 | ||
| Her2 | Negative | 16 | 6 | 0.80 |
| Positive | 11 | 3 | ||
| BIRADS score | II | 2 | 0 | 0.19 |
| IV | 6 | 1 | ||
| V | 8 | 7 | ||
ER: Oestrogen receptor, PR: Progesterone receptor, HER2: Human epidermal growth factor receptor 2, BIRADS: Breast imaging-reporting and database system
DISCUSSION
PET/CT proved its worth in many solid cancers. In breast cancer patients, its use was investigated both in assessing the local extent and distant metastatic disease. In assessing the local disease, MRI seems to be a better choice due to its higher resolution and better local detection.[8] PETCT is not a modality for the detection of local disease. Even for the detection of axillary disease, the sensitivity is <70%.[9] However, in most of the centres, it has become one of the initial modalities of investigation. In fact, there have been studies where it has shown to upstage the disease by 9–30%.[10] Groheux et al. detected distant metastasis in more than 10% of Stage IIB patients by PET/CT.[11] There were also studies which demonstrated that in patients with tumour size >2 cm, at least 14% of the patients were upstaged and nearly half of them had their treatment changed.[12] To have a better understanding of the role of WBPET as an initial investigation modality, this study was undertaken to determine its role in resource-limited Indian conditions.
The role of WBPET scan in breast cancer, especially in early breast cancer, has been heavily studied with most guidelines not recommending in its favour.[13] However, as already brought out, most breast surgeons routinely get a WBPET scan as a part of the initial staging workup. In our study, there were 13 patients out of 43 patients who had upstaging of the disease and 10 patients had whose treatment changed from curative to palliative intent after WBPET. Many of the doubtful liver lesions on ultrasound actually were detected to be metastatic deposits on WBPET. These lesions were histopathologically proven to be metastatic deposits of breast cancer.
As per Globocan 2018 report, breast cancer is the most commonly diagnosed cancer (27.7%) and is the leading cause of death (23.5%) among females in India. The median age of women at the time of diagnosis of breast cancer is approximately 61 years in the Western world, with the peak age being 60–70 years; however, in India, a higher proportion of patients with breast cancer tends to be premenopausal, and the peak age is between 40 and 50 years.[14] The median age of patients who were studied in our cohort was 49 years. This younger age of onset in Indian patients is a cause of concern as younger patients tend to have more aggressive diseases.
There has been increasing evidence to show the importance of young age, ER, PR status, and HER-2 expression in patients with breast cancers.[15] Although the numbers of patients studied were very small and it would be unreasonable to extrapolate this to the entire population of breast cancer, there was a definite trend toward more change in treatment in ER-negative patients (P = 0.35). Although it was not significant, the correlation of ER was still better than PR and Her2 Neu status.
All of our patients have undergone mammography before surgery or any other form of treatment. Of late, there are many data to detect a correlation between BIRADS staging and its association with histological subtypes, hormonal status, and also treatment outcome.[16] In experienced hands, the BIRADS system can be a very useful predictor of malignancy.[17]
To have a uniform reporting system for mammography, the American College of Radiology created BIRADS. It categorises breast lesion into six categories, one – denotes negative study, two – denotes benign lesion, three – denotes probably benign lesion, four – denotes suspicious abnormality, five – denotes lesion is highly suspicious of malignancy and six – denotes previously biopsy-proven malignancy. Two of our patients had either BIRADS IV or V at the time of diagnosis. Seven out of 15 of our BIRADS V patients had an upstage and change in treatment and one out of seven patients of BIRADS IV had a change of treatment on undergoing WB PET scan. Among all of the other variables, higher BIRADS seemed to be most related to change in treatment (P = 0.19)
Conventionally, the role of WBPET scan in breast cancer has been in the initial staging of advanced/suspected metastatic disease and to monitor drug response in patients. However, now, there have been various studies to correlate the outcome of WBPET scan with disease histology, grade, or even receptor status.[18]
It is known that ER-negative tumour has higher SUV uptake than ER-positive tumours.[19] Moreover, Grade III tumours have higher FDG avidity than Grade I tumour.[20] However, to date, these data in itself do not make any change in the treatment of breast cancer patients. To add-on to this vital question of whether we can have a SUV cutoff to suggest which disease will metastasise, we analysed our data. The ROC curve for the ability of the SUV of the primary lesion to identify patients requiring a change of treatment after PET-CT. The AUC = 0.639. At SUV of 7.25, sensitivity and specificity are 100% and 50%, respectively [Graph 2]. As per our data, a SUV of >7.2 of the primary breast lesion was suggestive that the probability of metastatic disease was high.
Limitations of the study
The biggest limitation of the study was the sample size. Breast cancer happens to be the most common cancer among females in India. To have a very significant outcome, a bigger sample size would have been ideal. However, in a resource-limited setting, getting WBPET scan for all patients with breast cancer was a formidable task.
Compliance with ethical standards
The Institutional Ethical Committee approved the study protocol and the study was conducted according to the Helsinki Declaration. Written consent was obtained from all study participants.
CONCLUSION
The use of 18F-FDG PET/CT has been shown to bring change in treatment in almost 20% of patients with breast cancer. Although the role of other variables in breast cancer such as hormone status, SUV of the primary lesion, and grade of lesion on BIRADS has also shown some association with change of treatment. However, one needs to have a study with a larger sample to convincingly answer these questions. As of now in primary breast cancer, 18F-FDG PET/CT cannot solely replace the conventional diagnostic procedure in primary breast cancer, with the potential to be useful as an additional imaging tool for the staging and management of the disease.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Breast Cancer: Prevention and Control. 2013. Geneva: World Health Organization; Available from: https://www.who.int/cancer/detection/breastcancer/en/index1.html [Last accessed on 2013 Dec 12]
- [Google Scholar]
- Epidemiology of breast cancer in Indian women. Asia Pac J Clin Oncol. 2017;13:289-95.
- [CrossRef] [PubMed] [Google Scholar]
- Is breast cancer the same disease in Asian and Western countries? World J Surg. 2010;34:2308-24.
- [CrossRef] [PubMed] [Google Scholar]
- Preoperative staging of large primary breast cancer with [18F] fluorodeoxyglucose positron emission tomography/computed tomography compared with conventional imaging procedures. J Clin Oncol. 2008;26:4746-51.
- [CrossRef] [PubMed] [Google Scholar]
- Role of maximum standardized uptake value in fluorodeoxyglucose positron emission tomography/computed tomography predicts malignancy grade and prognosis of operable breast cancer: A multi-institute study. Breast Cancer Res Treat. 2013;141:269-75.
- [CrossRef] [PubMed] [Google Scholar]
- FDG PET, PET/CT, and breast cancer imaging. Radiographics. 2007;27:S215-29.
- [CrossRef] [PubMed] [Google Scholar]
- Comparison of FDG PET and MRI for evaluating the tumor extent of breast cancer and the impact of FDG PET on the systemic staging and prognosis of patients who are candidates for breast-conserving therapy. Breast Cancer. 2009;16:97-104.
- [CrossRef] [PubMed] [Google Scholar]
- FDG-PET/CT in the staging of local/regional metastases in breast cancer. Breast. 2011;20:491-4.
- [CrossRef] [PubMed] [Google Scholar]
- Impact of FDG PET on the preoperative staging of newly diagnosed breast cancer. Eur J Nucl Med Mol Imaging. 2008;35:475-83.
- [CrossRef] [PubMed] [Google Scholar]
- Prognostic impact of (18) FDG-PET-CT findings in clinical stage III and IIB breast cancer. J Natl Cancer Inst. 2012;104:1879-87.
- [CrossRef] [PubMed] [Google Scholar]
- Preoperative PET/CT in early-stage breast cancer. Ann Oncol. 2012;23:2277-82.
- [CrossRef] [PubMed] [Google Scholar]
- Recommendations on the use of 18F-FDG PET in oncology. J Nucl Med. 2008;49:480-508.
- [CrossRef] [PubMed] [Google Scholar]
- Breast cancer care in India: The current scenario and the challenges for the future. Breast Care (Basel). 2008;3:21-7.
- [CrossRef] [PubMed] [Google Scholar]
- Breast cancer outcome and predictor of outcome: Are there age differentials? J Natl Cancer Inst Monogr. 1994;16:35-42.
- [Google Scholar]
- Correlation between BIRADS scoring and histology in women undergoing breast surgery in our institution. N Indian J Surg. 2019;10:25-30.
- [CrossRef] [Google Scholar]
- American society of clinical oncology/college of American pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol. 2010;28:2784-95.
- [CrossRef] [PubMed] [Google Scholar]
- Association between [18F] fluorodeoxyglucose uptake and prognostic parameters in breast cancer. Br J Surg. 2009;96:166-70.
- [CrossRef] [PubMed] [Google Scholar]
- Clinicopathological and prognostic relevance of uptake level using 18F-fluorodeoxyglucose positron emission tomography/computed tomography fusion imaging (18F-FDG PET/CT) in primary breast cancer. Jpn J Clin Oncol. 2008;38:250-8.
- [CrossRef] [PubMed] [Google Scholar]






